Associate Professor, Universidade Fernando Pessoa
| Other metabolic pathways: |
Blood glucose levels are kept at approximately constant levels around 4-5 mM. Glucose enters cells by facilitated diffusion. Since this process does not allow the cell to contain glucose at a higher concentration than the one present in the bloodstream, the cell (through the enxyme hexokinase) chemically modifies glucose by phosphorylation:
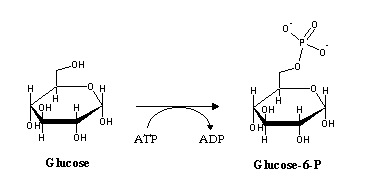
Since the cell membrane is impermeable to glucose-6-phosphate, this process effectively "traps" glucose inside the cell, allowing the recovery of more glucose from the bloodstream. Glucose-6-phosphate will be used in glycogen synthesis (a storage form of glucose) , production of other carbon compounds by the pentose-phosphate pathway, or degraded in order to produce energy- glycolysis.
In order to be used for energy production, glucose-6-phosphate must first be isomerized in fructose-6-phosphate. Fructose-6-phosphate is again phosphorylated to fructose-1,6-bisphosphate, in a reaction catalyzed by phosphofructokinase. This is the commited step of this metabolic pathway: from the moment glucose is transformed into fructose-1,6-bisphosphate it must proceed through glycolysis.
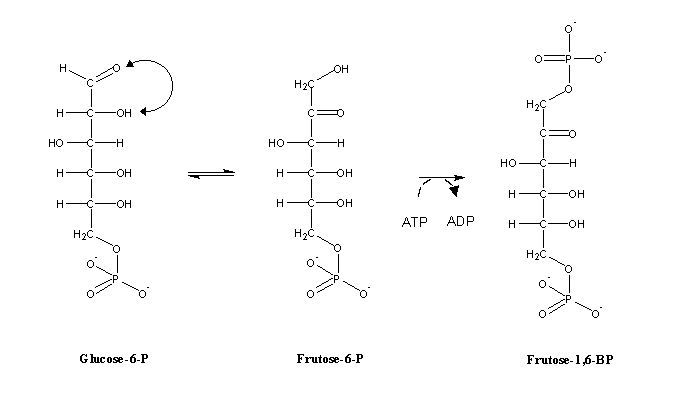
After this conversion, an inverse aldolic addition cleaves fructose-1,6-bisphosphate in two three-carbon molecules :
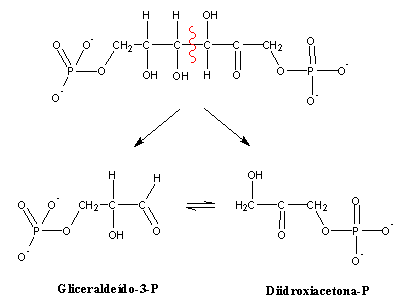
Both molecules (dihydroxyacetone phosphate and glyceraldehyde-3-phosphate) can easily be interconverted by isomerization. A single metabolic pathway is therefore enough to degrade both. This is why glucose-6-P was first isomerized to fructose-6-P: glucose-6-P breakdown through an inverse aldol addition would yield two quite different molecules (of two and four carbons, respectively), which would have to be degraded through two different pathways.
Aldehydes have very low redox potentials(around -600 to -500 mV). Oxidation of glyceraldehyde-3-phosphate by NAD+ (E0=-320 mV) is therefore quite spontanteous. Indeed, it is so exergonic that it can be used to produce ATP (ATP production from ADP and Pi can be performed if coupled to a two-electron redox reaction with a potential difference of at least 160 mV). ATP production happens through two consecutive steps: in the first step, gliceraldehyde-3-phosphate oxidation to a carboxylic acid is coupled to the phosphorylation of the produced carboxylic acid.
Phosphorilated acids (as well as phosphoenols and phosphoguanidines) contain very energetic phosphate groups: hydrolysis of these phosphate groups yields with very significant resonance stabilization. Therefore, the phosphate group attached to carbon 1 in 1,3-bisphosphoglycerate can be easily transferred to ADP, in order to produce ATP.
3-Phosphoglycerate is isomerized to 2-phosphoglycerate, which after dehydration (i.e. losing H2O) yields a phosphoenol:
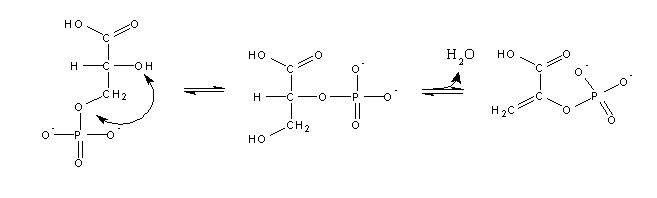
Due to its high phosphate transfer potential phosphoenolpyruvate can transfer a phosphate group to ADP:
Two ATP molecules are used in glycolysis, and four ATP are produced. NAD+ must be continuously regenerated, otherwise glycolysis will stop, since NAD+ is a substrate in one of the reactions. Under aerobic conditions, NADH transfers its two electrons to the electron-transport chain . In animal cells, in the absence of O2 NADH transfers its electrons to the end-product of glycolysis (pyruvate), yielding lactate. This is called fermentation : an internally balanced degradation, i.e., a process that uses one of its products as the final acceptor of the electrons it releases.
 |
Biochemistry,
by Donald Voet & Judith Voet An excellent text. It presents Biochemistry with frequent references to organic chemistry and biochemical logic. Highly reccommended for students of Biochemistry, Chemistry and Pharmaceutical Sciences. |
 |
Biochemistry,
Stryer A widely used classical text, frequently updated and re-issued. |
 |
Textbook
of Biochemistry with Clinical Correlations, Thomas Devlin Strongly advised to students in Nursing, Medicine, Dentistry, etc. Plenty of examples of application of biochemical knowledge to clinical cases. |
 |
Principles
of Biochemistry, Lehninger A widely used classical text, frequently updated and re-issued. |